What are salts?: Difference between revisions
| Line 72: | Line 72: | ||
==[[Salt properties]] == | ==[[Salt properties]] == | ||
Salts have certain properties that help to explain their behaviour in solution and their "damaging" effect on objects. | Salts have certain properties that help to explain their behaviour in solution and their "damaging" effect on objects. The most important are: | ||
*[[solubility]] | *[[solubility]] | ||
Revision as of 08:59, 20 September 2011
<bibimport />
Autoren: Hans-Jürgen Schwarz, NN
back to Fundamentals
Abstract[edit]
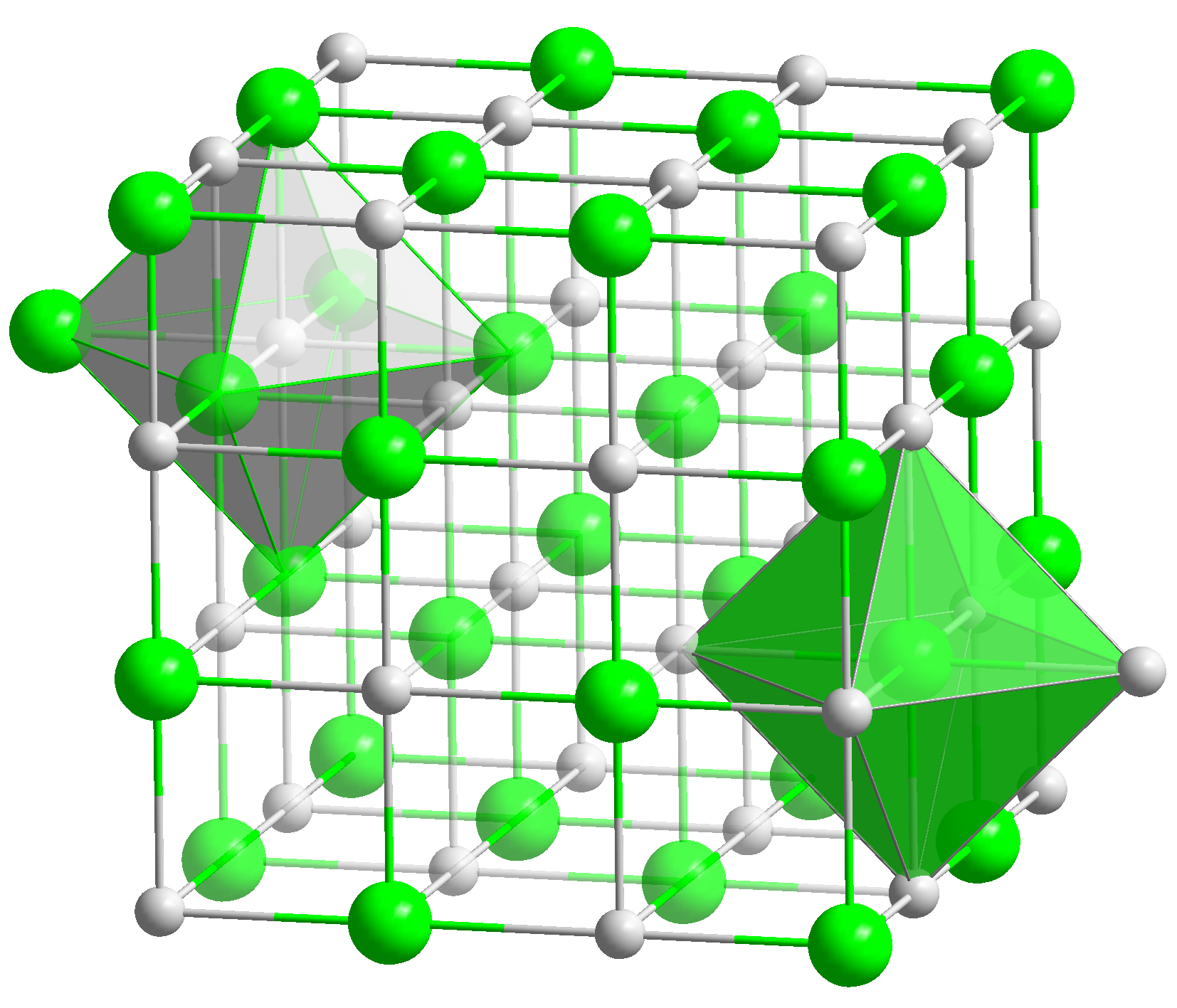
Salts are compounds made up of cations (e.g., K+, Ca2+, NH4+) and an anion, such as chloride (Cl-), nitrate (NO3-, that are held together by ionic bonds. They are crystalline materials.
The formation of salts[edit]
Salts[1] normally consist of positively charged ions, cations, and negatively charged ions, anions, that form a crystal lattice. In addition, some salts may include the water molecule (H2O) in the lattice. there are referred to as hydrated salts.
Salts are often the result of a neutralisation reaction, i.e., the mixture of an acid with a base.
Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle Na^+ + OH^- + H_3O^+ + Cl^- \rightarrow NaCl + 2H_2O}
base + acid → salt + water
While some salts, such as NaCl are neutral, that is, their solution does not change the normal pH 7 of water, other salts may be alkaline or acidic, depending on the strength of the participating acids and bases:
1. strong acid + strong base → neutral salt + H2O
2. strong acid + weak base → acidic salt + H2O
3. weak acid + strong base → alkaline salt + H2O
e.g.
1. Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle H_3O^+ + Cl^- + Na^+ + OH^- \rightarrow NaCl + H_2O }
2. Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle H_3O^+ + Cl^- + NH_3 \rightarrow NH_4Cl + H_2O }
3. Failed to parse (SVG (MathML can be enabled via browser plugin): Invalid response ("Math extension cannot connect to Restbase.") from server "https://wikimedia.org/api/rest_v1/":): {\displaystyle H_3O^ + HCO_3^- + 2Na^+ + 2OH^- \rightarrow Na_2CO_3 + 3H_2O }
The chemical notation of salts is written like this:
(positive ion (cation))x (negative ion (anion))y • nH2O; e.g. CaCl2 • 6 H2O ,
x,y,n being the number of the ions or water molecules
| Salts consist of cations and anions that are bound together with ionic bonds. They are crystalline materials. |
Composition of salts?[edit]
The most "damaging" and frequently found salts consist of these
anions:
- Sulphate SO42-
- Nitrate NO3-
- Chloride Cl-
- Carbonate CO32-
And the cations
- Sodium Na+
- Potassium K+
- Calcium Ca2+
- Magnesium Mg2+
other anions are:
- Acetate CH3COO-
- Formiate HCOO-
- Oxalate C2O42-
- Phosphate PO43-
further cations are:
- Ammonium NH4+
Salt properties[edit]
Salts have certain properties that help to explain their behaviour in solution and their "damaging" effect on objects. The most important are: