Calicum nitrate
Author: Amelie Stahlbuhk
back to Nitrate
| Calicum nitrate | |
| Mineralogical name | Calcium nitrate |
| Chemical name | Calcium nitrate |
| Trivial name | |
| Chemical formula | Ca(NO3)2 |
| Other forms | Ca(NO3)2•2H2O (Calcium nitrate dihydrate) Ca(NO3)2•3H2O (Calcium nitrate trihydrate) Ca(NO3)2•4H2O (Nitrocalcite) |
| Crystal system | |
| Crystal structure | |
| Deliquescence humidity 20°C | |
| Solubility (g/l) at 20°C | |
| Density (g/cm³) | 2.483 g/cm3 |
| Molar volume | 66.09 cm3/mol |
| Molar weight | 164.09 g/mol |
| Transparency | |
| Cleavage | |
| Crystal habit | |
| Twinning | |
| Phase transition | |
| Chemical behavior | |
| Comments | |
| Crystal Optics | |
| Refractive Indices | |
| Birefringence | |
| Optical Orientation | |
| Pleochroism | |
| Dispersion | |
| Used Literature | |
| [Robie.etal:1978]Title: Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar pressure and higher temperatures Author: Robie R.A., Hemingway B.S.; Fisher J.A. 
| |
Abstract[edit]
In this page, the calcium nitrate salt is discussed. Its behavior regarding solubility and hygroscopicity will be reviewed as well as its different hydration states.
Solubility[edit]
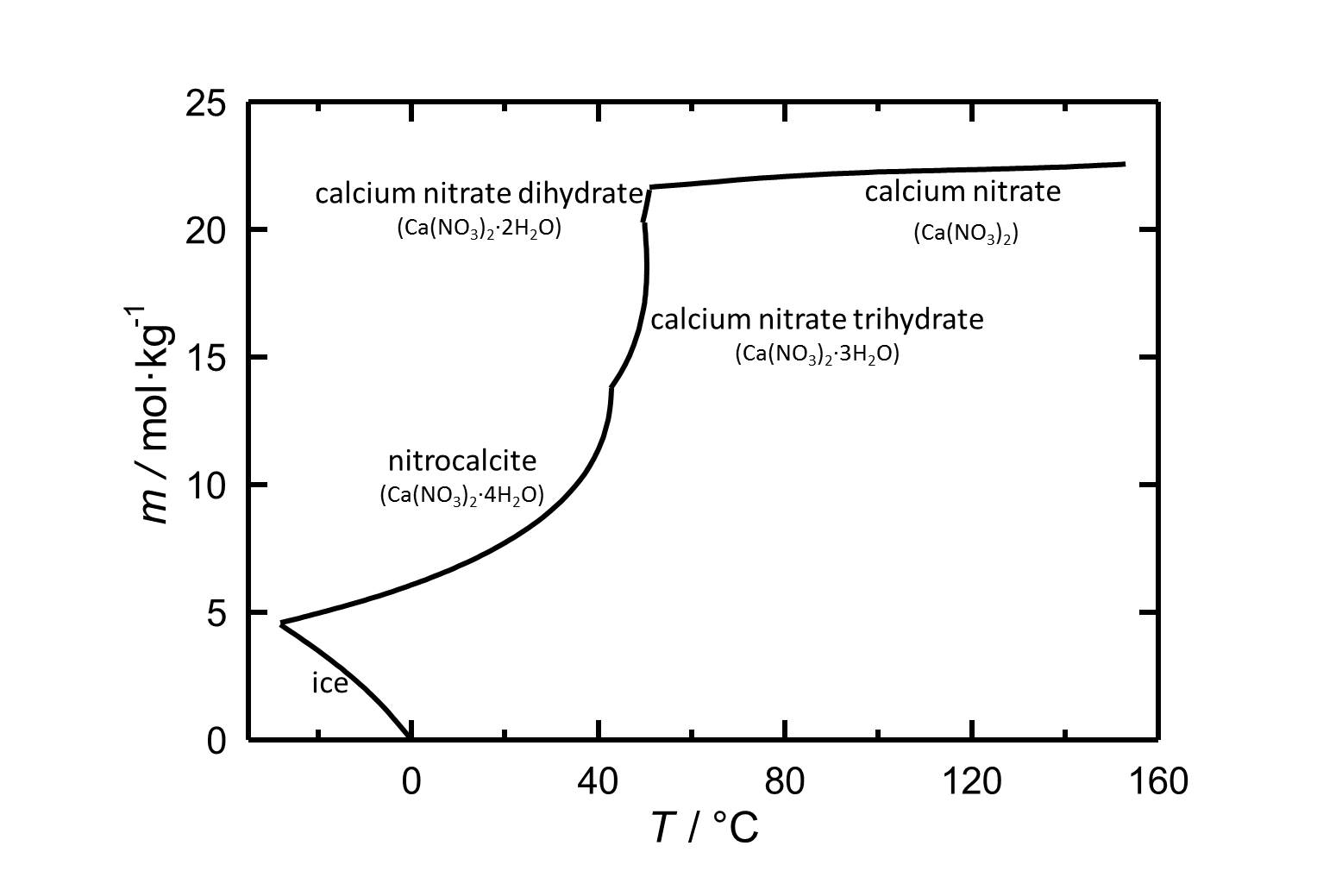
Under standard conditions the tetrahydrate of calcium nitrate Nitrocalcite is the stable phase. With its relatively high solubility in water it is a highly soluble salt. The temperature dependence of the solubility is shown in the solubility diagram, where in parts the solubility increases extremely with increasing temperature. The dehydrations to the Trihydrate, Dihydrate and at least to the anhydrous calcium nitrate take place at 43 °C, 49.5 °C and 51 °C, respectively.
Hygroscopcity[edit]
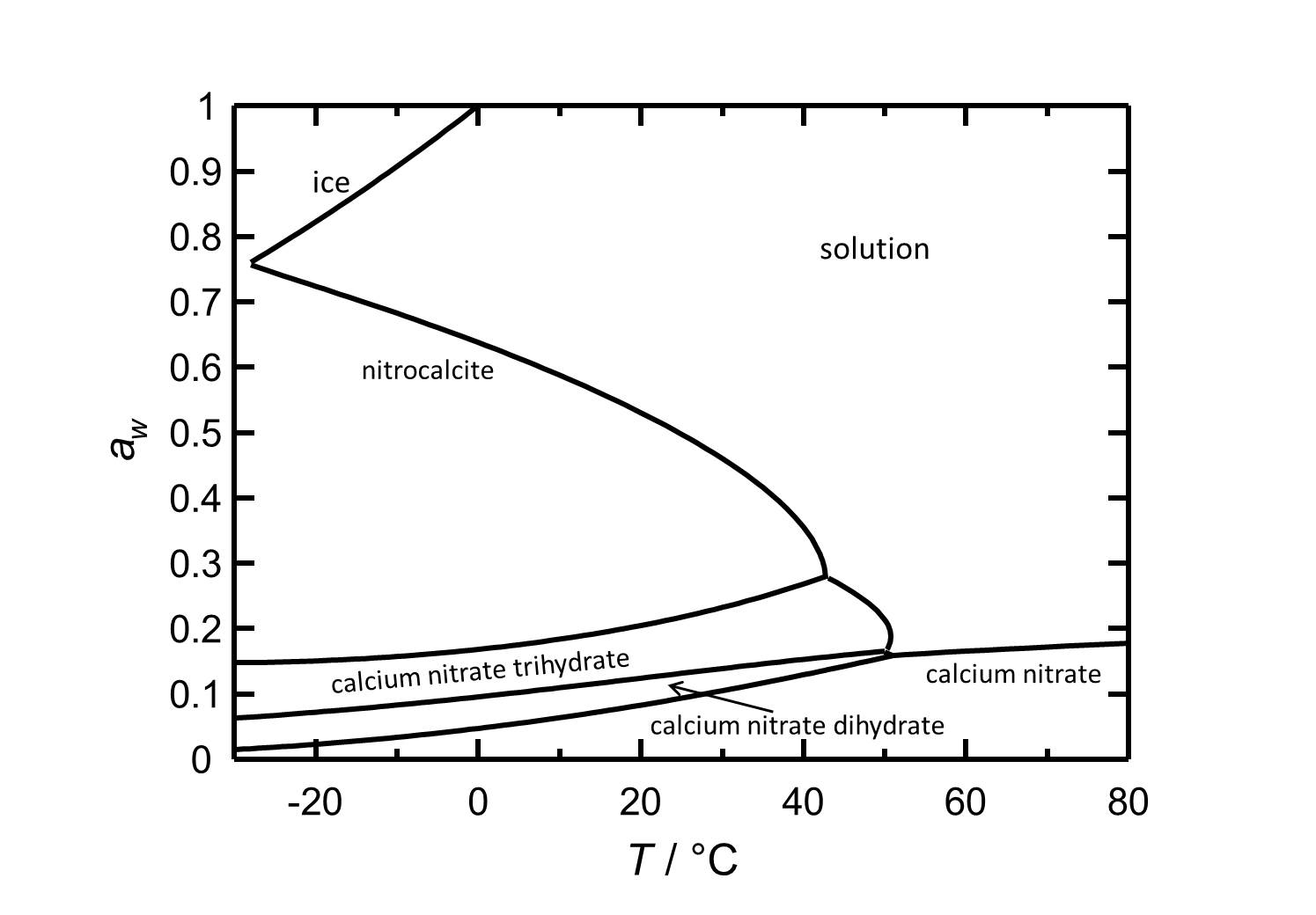
Calcium nitrate and its phases are hygroscopic salts. At a temperature of 25 °C the Nitrocalcite has a deliquescence humidity of about 50 %. The phase transitions to the other two hydrated forms and to the anhydrous calcium nitrate occur at relative humidities of 22 %, 13 % and 9 %, respectively.
References[edit]
Literature[edit]
| [Robie.etal:1978] | Robie R.A., Hemingway B.S.; Fisher J.A. (1978): Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar pressure and higher temperatures. In: U.S. Geol. Surv. Bull, 1452 () |  |