Antarcticite: Difference between revisions
Jump to navigation
Jump to search
No edit summary |
No edit summary |
||
| Line 9: | Line 9: | ||
|Crystal_System =trigonal | |Crystal_System =trigonal | ||
|Crystal_Structure = | |Crystal_Structure = | ||
|Deliqueszenzhumidity =30,8 | |Deliqueszenzhumidity =33,7% (10°C), 30,8% (20°C), 22,4% (30°C) | ||
|Solubility = | |Solubility = 5360 g/l | ||
|Density =1,68 g/cm³ | |Density =1,68 g/cm³ | ||
|MolVolume =128,0 cm<sup>3</sup>/mol | |MolVolume =128,0 cm<sup>3</sup>/mol | ||
| Line 18: | Line 18: | ||
|Crystal_Habit = | |Crystal_Habit = | ||
|Twinning = | |Twinning = | ||
|Refractive_Indices =n<sub>o</sub> =1,417<br> n<sub>e</sub> = 1,393 | |Refractive_Indices =n<sub>o</sub> =1,417-1,494<br>n<sub>e</sub> = 1,393-1,550 | ||
|Birefringence =Δ= 0,024 | |Birefringence =Δ= 0,024 | ||
|optical_Orientation = | |optical_Orientation = | ||
| Line 25: | Line 25: | ||
|Phase_Transition = | |Phase_Transition = | ||
|chemBehavior = | |chemBehavior = | ||
|Comments = | |Comments =hygroscopic and deliquescent | ||
}} | }} | ||
Revision as of 10:13, 7 July 2011
| Antarcticite[1][2] | |
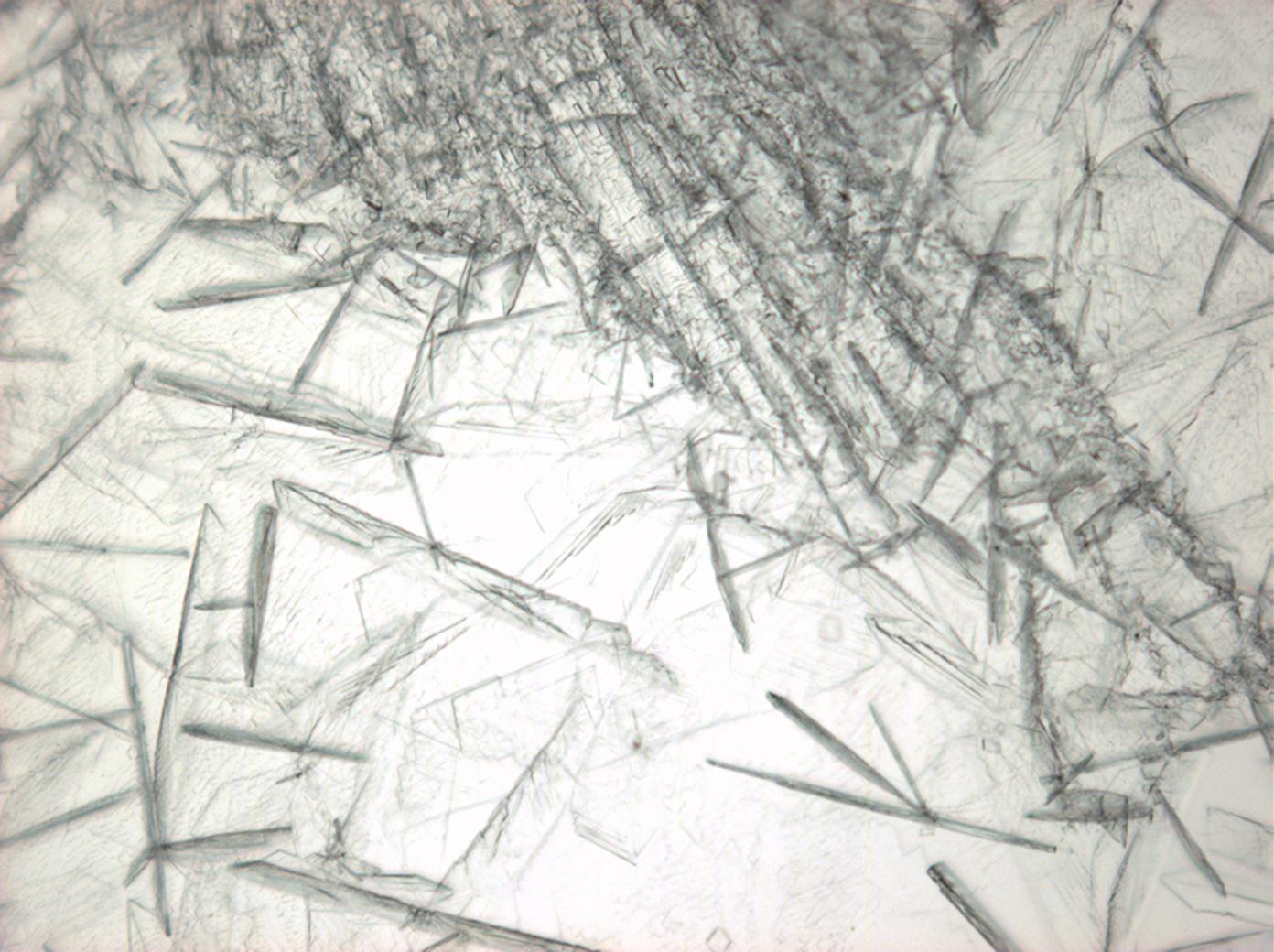
| |
| Mineralogical name | Antarcticite |
| Chemical name | Calciumchloride hexahydrate |
| Trivial name | |
| Chemical formula | CaCl2•6H2O |
| Other forms | |
| Crystal system | trigonal |
| Crystal structure | |
| Deliquescence humidity 20°C | 33,7% (10°C), 30,8% (20°C), 22,4% (30°C) |
| Solubility (g/l) at 20°C | 5360 g/l |
| Density (g/cm³) | 1,68 g/cm³ |
| Molar volume | 128,0 cm3/mol |
| Molar weight | 219,08 g/mol |
| Transparency | transparent |
| Cleavage | perfect |
| Crystal habit | |
| Twinning | |
| Phase transition | |
| Chemical behavior | |
| Comments | hygroscopic and deliquescent |
| Crystal Optics | |
| Refractive Indices | no =1,417-1,494 ne = 1,393-1,550 |
| Birefringence | Δ= 0,024 |
| Optical Orientation | |
| Pleochroism | |
| Dispersion | |
| Used Literature | |
| {{{Literature}}} | |
back to Chloride
Weblinks[edit]
- ↑ http://webmineral.com/data/Antarcticite.shtml viewed on 29/07/2010
- ↑ http://www.mindat.org/min-251.html viewed on 29/07/2010