Calcium chloride: Difference between revisions
| (24 intermediate revisions by 3 users not shown) | |||
| Line 3: | Line 3: | ||
back to [[Chloride]] | back to [[Chloride]] | ||
==Abstract== | ==Abstract== | ||
The different | The different hydrates of calcium chloride are presented, as well as their behavior regarding solubility and hygroscopicity. | ||
==Hydrate stages== | ==Hydrate stages== | ||
[[Sinjarite]]: CaCl<sub>2</sub>•2H<sub>2</sub>O <br> [[Calcium chloride tetrahydrate]]: CaCl<sub>2</sub>•4H<sub>2</sub>O <br> [[Antarcticite]]: CaCl<sub>2</sub>•6H<sub>2</sub>O | [[Calcium chloride monohydrate]]: CaCl<sub>2</sub>•H<sub>2</sub>O <br> [[Sinjarite]]: CaCl<sub>2</sub>•2H<sub>2</sub>O <br> [[Calcium chloride tetrahydrate]]: CaCl<sub>2</sub>•4H<sub>2</sub>O <br> [[Antarcticite]]: CaCl<sub>2</sub>•6H<sub>2</sub>O | ||
==Solubility== | ==Solubility== | ||
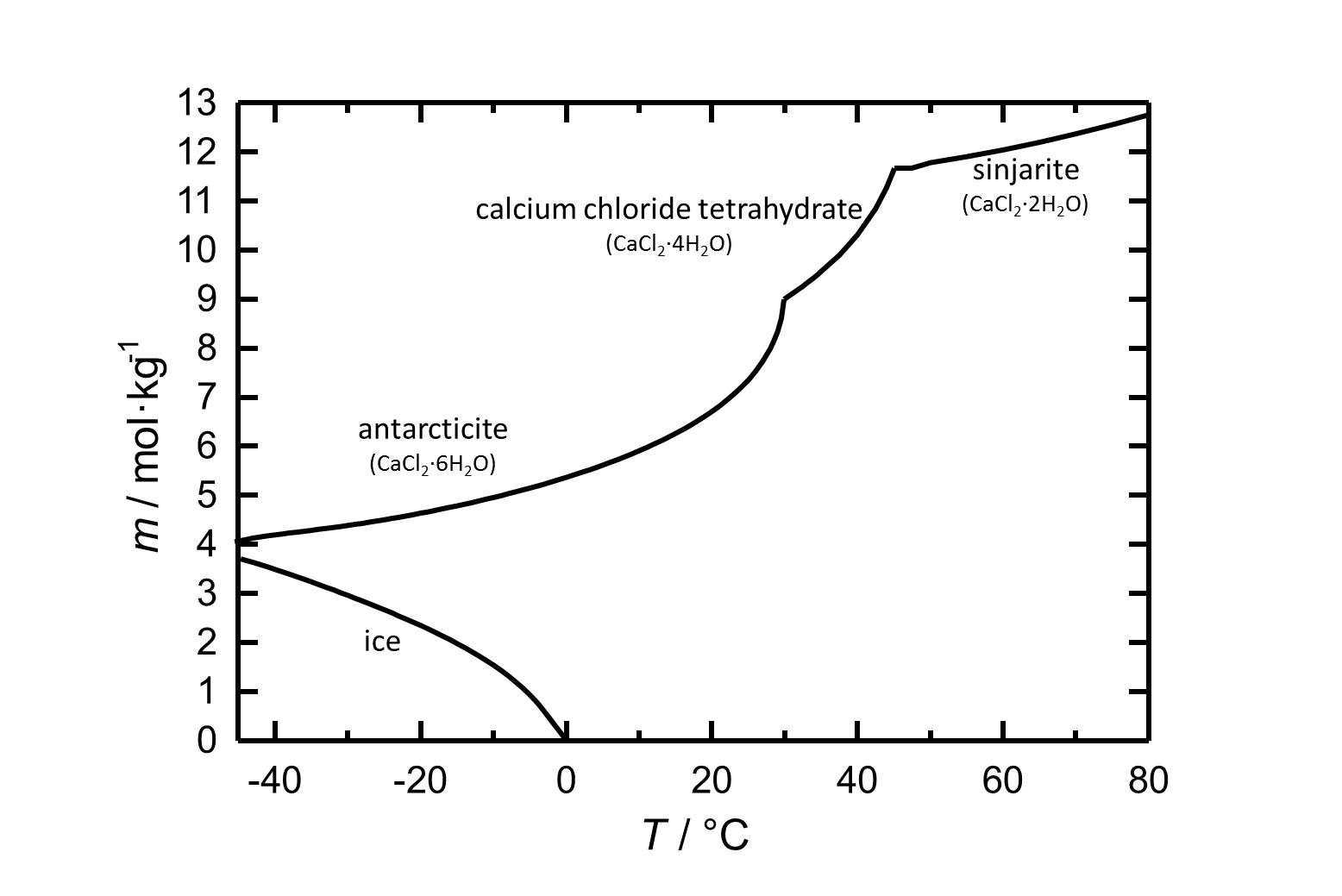
[[ | [[file:L CaCl2 d.jpg|thumb|left|800px|Figure 1: Solubility of calcium chloride in water. The molality ''m'' [n(CaCl<sub>2</sub>•xH<sub>2</sub>O)•kg(H<sub>2</sub>O)<sup>-1</sup>] is plotted against the temperature.]] | ||
<br clear=all> | |||
Under standard conditions the hexahydrate of calcium chloride [[Antarcticite]] is the stable form. The salt has got a high solubility in water which increases with increasing temperatures. The dehydration steps to the [[calcium chloride tetrahydrate]] and to [[sinjarite]] take place at temperatures of 30 °C and 45 °C, respectively. | |||
==Hygroscopicity== | ==Hygroscopicity== | ||
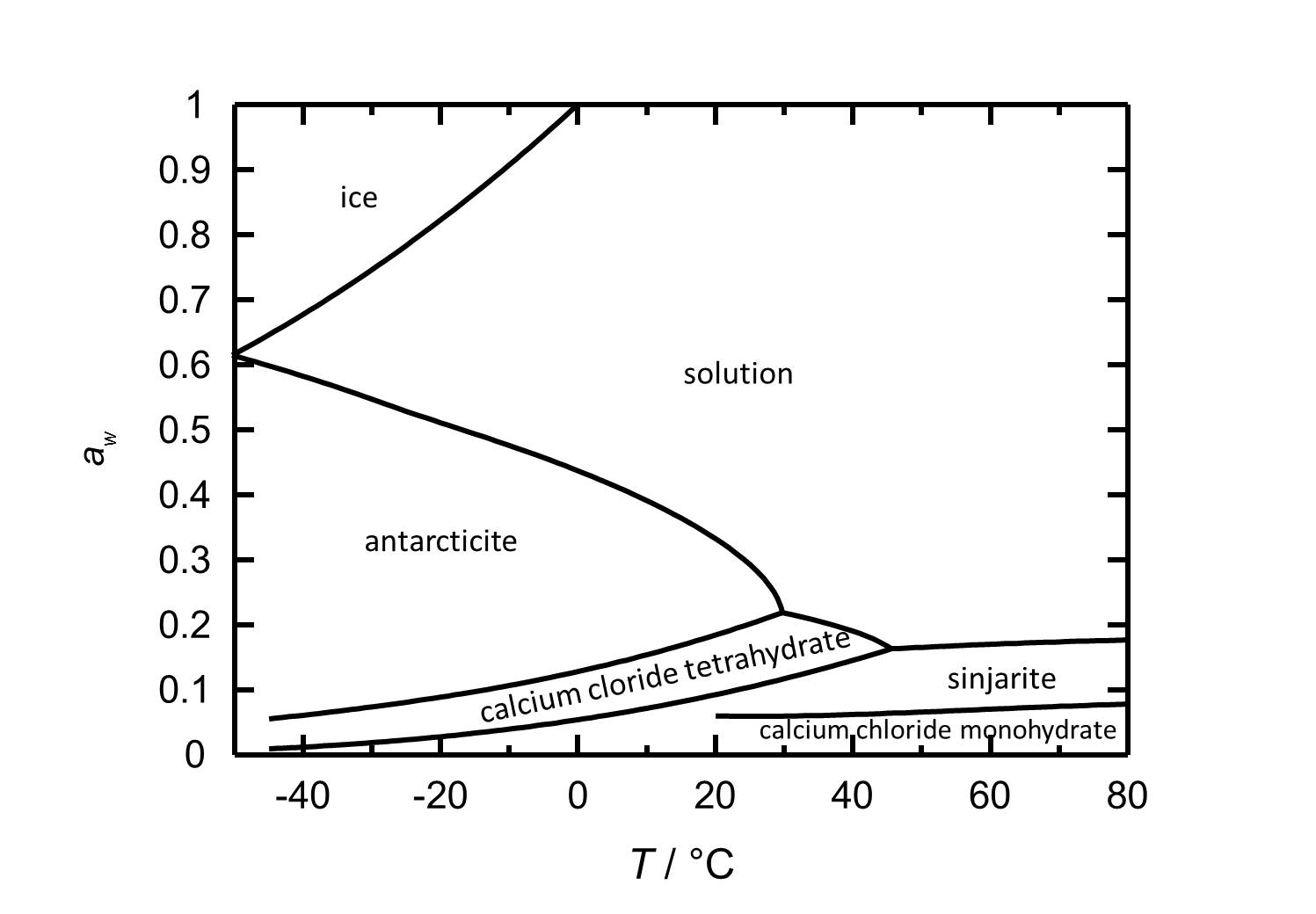
[[file:D CaCl2 e.jpg|thumb|left|800px|Figure 2: Deliquescence behaviour of calcium chloride in the temperature range from -45 to 80 °C. The water activity ''a<sub>w</sub>'' is plotted against the temperature.]] | |||
<br clear=all> | |||
The hydration/dehydration and crystallization/deliquescence processes in the CaCl<sub>2</sub>-H<sub>2</sub>O system can occur by either changing relative humidity or temperature. The deliquescence relative humidity decreases with increasing temperature. At room temperature (20ºC), the hexahydrate [Antarcticite]] is the stable phase, having a deliquescence humidity of about 30% RH. Lowering the relative humidity at the same temperature, calcium chloride tetrahydrate forms at values below 18% RH, while dehydration to the dihydrate ([[sinjarite]]) occurs at 9% RH, while the latter changes to the monohydrate at 6% RH. | |||
<br clear="all"> | |||
{|border="2" cellspacing="0" cellpadding="4" width="30%" align="left" class="wikitable" | |||
|+''Table 1: Deliquescence and equilibrium humidities at 20 °C.'' | |||
|- | |||
|bgcolor = "#F0F0F0" align=center| '''Phase transition''' | |||
|bgcolor = "#F0F0F0" align=center| '''Deliquescence or equilibrium humidity at 20°C''' | |||
|- | |||
|bgcolor = "#FFFFEO" align=center| [[Antarcticite]]-solution | |||
|bgcolor = "#FFFFEO" align=center| 33.3 % | |||
|- | |||
|bgcolor = "#FFFFEO" align=center| [[Antarcticite]]-[[Calcium chloride tetrahydrate]] | |||
|bgcolor = "#FFFFEO" align=center| 18.5 % | |||
|- | |||
|bgcolor = "#FFFFEO" align=center| [[Calcium chloride tetrahydrate]]-[[Sinjarite]] | |||
|bgcolor = "#FFFFEO" align=center| 9 % | |||
|- | |||
|bgcolor = "#FFFFEO" align=center| [[Sinjarite]]-[[Calcium chloride monohydrate]] | |||
|bgcolor = "#FFFFEO" align=center| 6 % | |||
|} | |||
<br clear="all"> | |||
==References== | ==References== | ||
| Line 25: | Line 53: | ||
[[Category:Stahlbuhk,Amelie]][[Category:Calcium chloride]] [[Category:Chloride]] [[Category:R-MSteiger]] [[Category: | [[Category:Stahlbuhk,Amelie]][[Category:Calcium chloride]] [[Category:Chloride]] [[Category:R-MSteiger]] [[Category:editing]] [[Category:Salt]] | ||
Latest revision as of 21:25, 30 August 2016
Author: Amelie Stahlbuhk
back to Chloride
Abstract[edit]
The different hydrates of calcium chloride are presented, as well as their behavior regarding solubility and hygroscopicity.
Hydrate stages[edit]
Calcium chloride monohydrate: CaCl2•H2O
Sinjarite: CaCl2•2H2O
Calcium chloride tetrahydrate: CaCl2•4H2O
Antarcticite: CaCl2•6H2O
Solubility[edit]
Under standard conditions the hexahydrate of calcium chloride Antarcticite is the stable form. The salt has got a high solubility in water which increases with increasing temperatures. The dehydration steps to the calcium chloride tetrahydrate and to sinjarite take place at temperatures of 30 °C and 45 °C, respectively.
Hygroscopicity[edit]
The hydration/dehydration and crystallization/deliquescence processes in the CaCl2-H2O system can occur by either changing relative humidity or temperature. The deliquescence relative humidity decreases with increasing temperature. At room temperature (20ºC), the hexahydrate [Antarcticite]] is the stable phase, having a deliquescence humidity of about 30% RH. Lowering the relative humidity at the same temperature, calcium chloride tetrahydrate forms at values below 18% RH, while dehydration to the dihydrate (sinjarite) occurs at 9% RH, while the latter changes to the monohydrate at 6% RH.
| Phase transition | Deliquescence or equilibrium humidity at 20°C |
| Antarcticite-solution | 33.3 % |
| Antarcticite-Calcium chloride tetrahydrate | 18.5 % |
| Calcium chloride tetrahydrate-Sinjarite | 9 % |
| Sinjarite-Calcium chloride monohydrate | 6 % |