Epsomite: Difference between revisions
(Created page with "Category:Epsomite") |
No edit summary |
||
| (54 intermediate revisions by 8 users not shown) | |||
| Line 1: | Line 1: | ||
[[Category:Epsomite]] | Authors: [[user:Hschwarz|Hans-Jürgen Schwarz]], [[user:TMueller|Tim Müller]], Nils Mainusch | ||
<br>English Translation by Sandra Leithäuser | |||
<br> | |||
back to [[Sulfate]] | |||
{{Infobox_Salt | |||
|Footnote =<ref>http://webmineral.com/data/Epsomite.shtml seen on 29.072010</ref><ref>http://www.mindat.org/min-1393.html seen on 29.072010</ref><ref><bib id="Steiger.etal:2011"></ref><ref><bib id="Lide:1995"></ref> | |||
|photo = [[File:HJS MgSO4 092503-2.jpg|300px]] | |||
|mineralogical_Name = Epsomite | |||
|chemical_Name = Magnesium sulfate heptahydrate | |||
|Trivial_Name = Bitter Salts, reichardtite, seelandite | |||
|chemical_Formula = MgSO<sub>4</sub>•7H<sub>2</sub>O | |||
|Hydratforms = [[Kieserite]] (MgSO<sub>4</sub>•H<sub>2</sub>O)<br>[[Sanderite]] (MgSO<sub>4</sub>•2H<sub>2</sub>O)<br>[[Starkeyite]] (MgSO<sub>4</sub>•4H<sub>2</sub>O)<br>[[Pentahydrite]](MgSO<sub>4</sub>•5H<sub>2</sub>O) <br>[[Hexahydrite]] (MgSO<sub>4</sub>•6H<sub>2</sub>O)<br>[[Meridianiite]] (MgSO<sub>4</sub>•11H<sub>2</sub>O)<br>[[Magnesium 12-Hydrate]] | |||
|Crystal_System = orthorhombic | |||
|Crystal_Structure = | |||
|Deliqueszenzhumidity = 91.3 % | |||
|Solubility = 2.893 mol/kg | |||
|Density = 1.68 g/cm<sup>3</sup> | |||
|MolVolume = 146.8 cm<sup>3</sup>/mol | |||
|Molweight = 246.48 g/mol | |||
|Transparency = transparent to translucent | |||
|Cleavage = clear to perfect | |||
|Crystal_Habit = small, acicular, fibre like crystals, granular agregates, crusts | |||
|Twinning = rare | |||
|Refractive_Indices = n<sub>x</sub> = 1.432<br>n<sub>y</sub> = 1.453<br>n<sub>z</sub> = 1.4609 | |||
|Birefringence = Δ = 0.0284 | |||
|optical_Orientation = biaxial negative | |||
|Pleochroism = none | |||
|Dispersion =51.1° | |||
|Phase_Transition = | |||
|chemBehavior = | |||
|Comments = can be produced from an aqueous solution under 50°C | |||
|Literature =<bib id="Steiger.etal:2011"/> <bib id="Lide:1995"/> <bib id="Dana:1951"/> | |||
}} | |||
== Introduction == | |||
== Occurrence of epsomite == | |||
The natural mineral epsomite was first extracted in 1695, from the mineral waters near Epsom (London), where the mineral name was derived from. Just as [[kieserite]] (magnesium sulfate mono hydrate), single crystals rarely occur in nature. Both magnesium salts, as well as their hydrate forms are extracted from alpine deposits and occur together with [[sylvite]] and [[halite|rock salt]] in north Germany, and in the top layer of salt deposits by Staßfurt (near Magdeburg). | |||
== Information on the origin and formation of epsomite on monuments == | |||
The formation of magnesium sulfate on monuments requires the presence of soluble magnesium compounds, and magnesium ions. Different building materials on historic structures can contain magnesium compounds. Some examples are listed below:<br> | |||
*'''Utilization of lime with dolomite components''': [[Dolomite]] (CaMg(CO<sub>3</sub>)<sub>2</sub> ) is a double salt. If lime stone containing dolomite components is heated, slaked and used as mortar, CaCO<sub>3</sub> and MgCO<sub>3</sub>·xH<sub>2</sub>O form through this process (e.g. [[Nesquehonite]] MgCO<sub>3</sub>·3H<sub>2</sub>O including the molecules from the water of crystallization). The water solubility of MgCO<sub>3</sub>·3H<sub>2</sub>O at 1,76 g/l, is significantly above the solubility of [[calcite]] (0,014 g/l) and [[dolomite]] (0,078 g/l). If magnesium ions have been dissolved by moisture, they can form several different magnesium salts in the presents of the corresponding anions. The formation of magnesium sulfate can occur if, in connection with dolomite containing plasters, [[gypsum]] was used for stucco or plaster, readily supplying sulfates. | |||
*'''Utilization of magnesia containing binder''': Magnesia binder consists essentially of MgO and MgCl<sub>2</sub> or magnesium sulfate. The fully cured and solidified binder can contain highly hygroscopic magnesium salts, which can be converted to magnesium sulfate. | |||
*'''Utilization of cement''': According to the German standard, DIN 1164 <bib id="DIN1164:1994"/> a maximum content MgO of 5 M.% is tolerated in cements. Apart from the possibility of causing damage by delayed quenching, due to the expansion of the magnesia, magnesium ions can theoretically be released and cause the formation of magnesium sulfate. | |||
Deicing salt can be another source of magnesium sulfate, it often contains a small proportion of readily soluble MgCl<sub>2</sub>. Furthermore, the magnesium supply can occur through constant soil leaching in connection with rising damp. | |||
<br> | |||
== Solubility properties == | |||
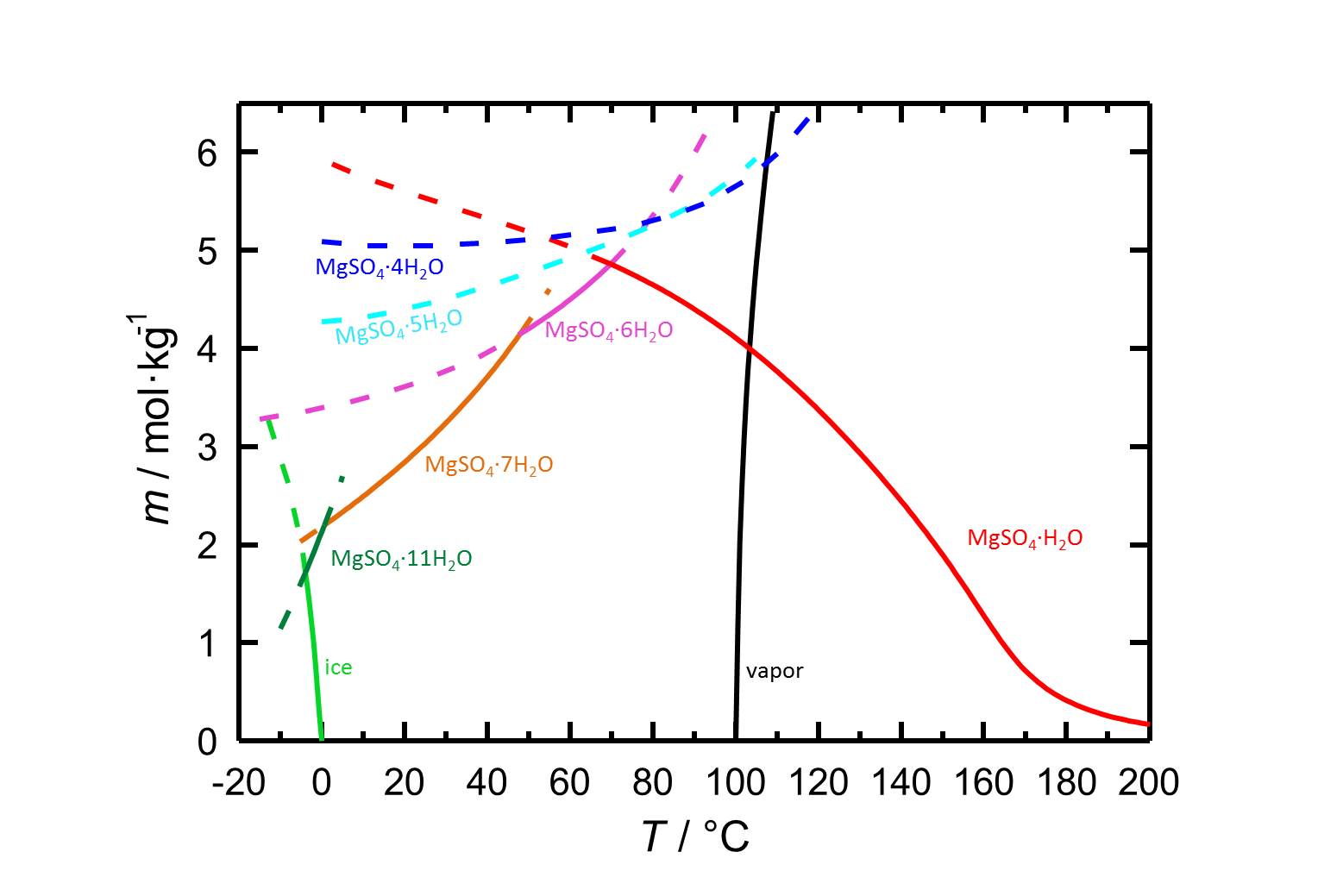
[[file:S MgSO4.jpg|thumb|800 px|left|'''Figure 1:''' Solubilities in the system MgSO<sub>4</sub>-H<sub>2</sub>O and its temperature dependence. The molality ''m'' [n(MgSO<sub>4</sub>•xH<sub>2</sub>O)•kg(H<sub>2</sub>O)<sup>-1</sup>] is plotted versus the temperature. Equilibria of the different hydrated forms can be distiguished by the different colours of the curves. Dotted lines represent the metastable equilibria. In addition to the solubilities, freezing and boiling points are given. According to <bib id="Steiger.etal:2011a"/>.]] | |||
<br clear=all> | |||
The solubility of the above magnesium sulfate forms is significantly above 100 g/l (20°C), and they are part of the group of easily soluble salts. In principle, this leads to a great mobility of the salt and frequent shifting of the accumulation zones inside the material fabric. Due to the influence of temperature on solubility, there is a risk of precipitation of the dissolved salts in combination with rapid temperature drops. | |||
<br clear=all> | |||
== Hygroscopicity == | |||
The low hygroscopicity of the pure salt epsomite which becomes evident in the high value of the equilibrium moisture content in the range of 88-90% RH, cannot be considered in isolation. In mixed systems, i.e. under the influence of foreign ions the sorption point is lower (see table). The possibility of hygroscopic moisture uptake with the connected difficulties is still possible despite the high [[deliquescence|Deliquescence]] point. <br> | |||
<br clear="all"> | |||
[[File:D MgSO4 e.jpg|thumb|800px|left|'''Figure 2:''' Deliquescence behaviour in the system MgSO<sub>4</sub>-H<sub>2</sub>O. The water activity ''a<sub>w</sub>'' is plotted versus the temperature. Different colours label equilibrium and deliquescence humidities of the different phases of the system. Dotted lines represent metastable equilibria. According to <bib id="Steiger.etal:2011a"/>.]] | |||
<br clear="all"> | |||
<br clear="all"> | |||
{|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable" | |||
|+''Table 1:Deliquescence humidities at different temperatures [according to <bib id="Steiger.etal:2011a"/>]'' | |||
|- | |||
|bgcolor = "#F0F0F0" align=center| 10°C | |||
|bgcolor = "#F0F0F0" align=center| 20°C | |||
|bgcolor = "#F0F0F0" align=center| 25°C | |||
|bgcolor = "#F0F0F0" align=center| 30°C | |||
|- | |||
|bgcolor = "#FFFFEO" align=center| 93.7% r.h. | |||
|bgcolor = "#FFFFEO" align=center| 91.3% r.h. | |||
|bgcolor = "#FFFFEO" align=center| 90.3% r.h. | |||
|bgcolor = "#FFFFEO" align=center| 89.1% r.h. | |||
|} | |||
<br clear="all"> | |||
===Moisture Sorption=== | |||
{|border="2" cellspacing="0" cellpadding="4" width="52%" align="left" class="wikitable sortable" | |||
|+''Tabele 2: Behavior concerning moisture sorption of magnesium sulfate [according to <bib id="Vogt.etal:1993"/>]'' | |||
|- | |||
|bgcolor = "#F0F0F0"| '''Moisture sorption at''' | |||
|bgcolor = "#F0F0F0"| '''87%r.F.''' | |||
|bgcolor = "#F0F0F0"| '''81%r.F.''' | |||
|bgcolor = "#F0F0F0"| '''70%r.F.''' | |||
|bgcolor = "#F0F0F0"| '''61%r.F.''' | |||
|bgcolor = "#F0F0F0"| '''50%r.F.''' | |||
|- | |||
|bgcolor = "#F7F7F7"| '''MgSO<sub>4</sub>''' | |||
|bgcolor = "#FFFFEO" align=center| 76 | |||
|bgcolor = "#FFFFEO" align=center| 75 | |||
|bgcolor = "#FFFFEO" align=center| 70 | |||
|bgcolor = "#FFFFEO" align=center| 71 | |||
|bgcolor = "#FFFFEO" align=center| 27 | |||
|- | |||
|bgcolor = "#F7F7F7"| '''MgSO<sub>4</sub> + NaCl'''<br>(1:1 molar mixture) | |||
|bgcolor = "#FFFFEO" align=center| 240 | |||
|bgcolor = "#FFFFEO" align=center| 146 | |||
|bgcolor = "#FFFFEO" align=center| 75 | |||
|bgcolor = "#FFFFEO" align=center| 50 | |||
|bgcolor = "#FFFFEO" align=center| 20 | |||
|} | |||
<br clear="all"> | |||
== Crystallization pressure == | |||
Due to the high solubility of the salt, dissolving and recrystallization processes take place under the appropriate humidity conditions. In theory the occurring crystallization pressure can be calculated and ranges between 10,5-12,5 N/mm<sup>2 </sup> for epsomite and between 11,8- 14,1 N/mm<sup>2</sup> for [[hexahydrite]]. Compared to other damaging salts these values are thus to be found in the lower third of a calculated scale of values, alltogether ranging from 7,2 bis 65,4 N/mm<sup>2</sup> [according to <bib id="Winkler:1975"/>]. | |||
== Hydration behavior == | |||
The system MgSO<sub>4</sub> – H<sub>2</sub>O: the listed hydrate stages of magnesium sulfates are known to be stable compounds. With the exception of magnesium sulfate- 12- hydrate, all the above stages of the chemically combined water of magnesium sulfate on monuments are visible, but essentially only epsomite, [[hexahydrite]], [[pentahydrite]] and [[kieserite]] occur. | |||
At room temperature and an RH between 50%-90%, epsomite is the most stable hydrate stage. If the RH drops significantly lower than 50% at room temperature, the chemically combined water is released and lower hydrate stages are formed. Hexahydrite (MgSO<sub>4</sub><sub></sub> × 6H<sub>2</sub>O) as pure salt is only stable at a temperature range of approx. 48°C to 67,5 °C. | |||
Pentahydrite has been thought to be metastable or unstable when exposed to the air, nevertheless the existence of these two salt phases has been detected by X-ray diffraction on building structures. The expulsion of the hydrate water can occur up to the formation of kieserite at elevated temperatures. | |||
== Hydration pressure == | |||
Possible variations in magnesium sulfate´s content of chemically combined water on constructions are a fact, and it is likely, that changes in the hydrate stages [[pentahydrite]], [[hexahydrite]] and epsomite (subject to climatic fluctuations) are taking place in situ. The addition of a water molecule into the crystal lattice of hexahydrite, resulting in the transformation to epsomite, is associated with an increase in volume of around 10%. At temperatures from 0-20°C and 70% RH the resulting hydration pressure can be specified at values between 6,8 –9,7 N/mm<sup>2</sup>. The conversion from kieserite to hexahydrite causes an increase in volume of approx. 140% [according to <bib id="Stark.etal:1996"/>]. | |||
== Conversion reaction == | |||
The following case is a good example for the potential damage connected with the formation of epsomite: if magnesium carbonate (as [[magnesite]]) is present in the material structure of a monument, epsomite can form through the influence of sulfuric acid. According to <bib id="Stark.etal:1996"/> this conversion is linked to an increase in volume of over 400%. | |||
<br> | |||
<br> | |||
== Analytical identification == | |||
On objects, crystallized magnesium sulfate can appear in differing morphologies, but specific forms appear more frequently. One examined object displayed a loose crust of an opaque, gray to yellow substance in situ, which was a mixture of epsomite and hexahydrite (part of the thesis by Mainusch <bib id= "Mainusch:2001"/>). At the monastery church St. Johann at Müstair epsomite appeared as “granular crusts”. Magnesium sulfate efflorescence in the shape of salt whiskers was detected at St. Georgs church in Steiermark (Styria). | |||
<!-- | |||
=== Micro- chemistry === | |||
--> | |||
=== Microscopy === | |||
'''Laboratory examination:''' | |||
Breathing onto a (predominantly) magnesium sulfate efflorescence, causes no observable macroscopic changes. However, characteristics are: the good solubility in water, a pH of 7, the formation of a ring-shaped slightly raised seam (visible by the naked eye), or a transparent layer that remains in place after the evaporation of the aqueous solvent. | |||
In laboratory tests on objects the results should be double checked with the microscope. Observations of the raw sample material and the recrystallized salt, show that magnesium sulfate crystals appear in unspecific forms in sample material. | |||
Experiments to produce well-shaped, single magnesium sulfate crystals, by recrystallization in aqueous solution have not been very successful, because of a strong tendency for intergrowth. Usually the previously mentioned, distinct, ring-shaped seam of intergrown crystals forms, when the magnesium sulfate containing solute is evaporated. Epsomite shows a low solubility in anhydrous ethanol and glycerin under the microscope. | |||
For further examinations using [[polarized light microscopy|polarized light microscopy]], a considerable difficulty is the creation of good preparations, due to the tendency of magnesium sulfate to form only few equidimensional or elongated single crystals. The low solubility in ethanol, nevertheless allows to isolate single particles by repeatedly adding recrystallized material. Both on the base material as well as the recrystallized product, examination using polarized light microscopy should be carried out.<br> | |||
'''Refractive indices''': n<sub>X</sub> = 1,433, n<sub>y</sub> = 1,455, n<sub>z</sub> <br> | |||
'''Birefringence: '''Δ = max. 0,028 <br> | |||
'''Crystal class:''' orthorhombic | |||
<br> | |||
<br> | |||
'''[[Polarized light microscopy|Polarized light microscopic examination:]]'''<br> | |||
The allocation of epsomite to the refractive indices is carried out according to the immersion method (successively by means of embedding media n<sub>D</sub>=1,518; n<sub>D</sub>=1,47; n<sub>D</sub>=1,46). When using an immersion medium with a refractive index of n<sub>D</sub>=1,45 on many single particles a slight but distinct variation in relief is visible at rotation. Because epsomite belongs to the class of orthorhombic crystals, it never appears in an oblique but always in a parallel symmetrical extinction. Beside epsomite, hexahydrite often forms, which is monoclinic and has nearly identical refractive indices. A distinction between these two hydrate stages of magnesium sulfate is only possible through the allocation of the crystal class. Due to the low path difference epsomite crystals usually only show low inference colors in the range of the first order. | |||
'''Possibility for mistakes:'''<br> | |||
Epsomite/[[hexahydrite]] are to be allocated, once the examination criteria below have been clarified: | |||
*good water solubility | |||
*characteristic appearance at recrystallization | |||
*low birefringence | |||
<br> | |||
<!-- | |||
== X- Ray diffraction == | |||
== Raman-Spectroscopiy == | |||
== DTA / TG == | |||
== IR-Spectroscopy == | |||
= <br>Umgang mit Epsomitschäden = | |||
--> | |||
== Salts and deterioration pattern == | |||
=== On objects === | |||
=== Under the polarising microscope === | |||
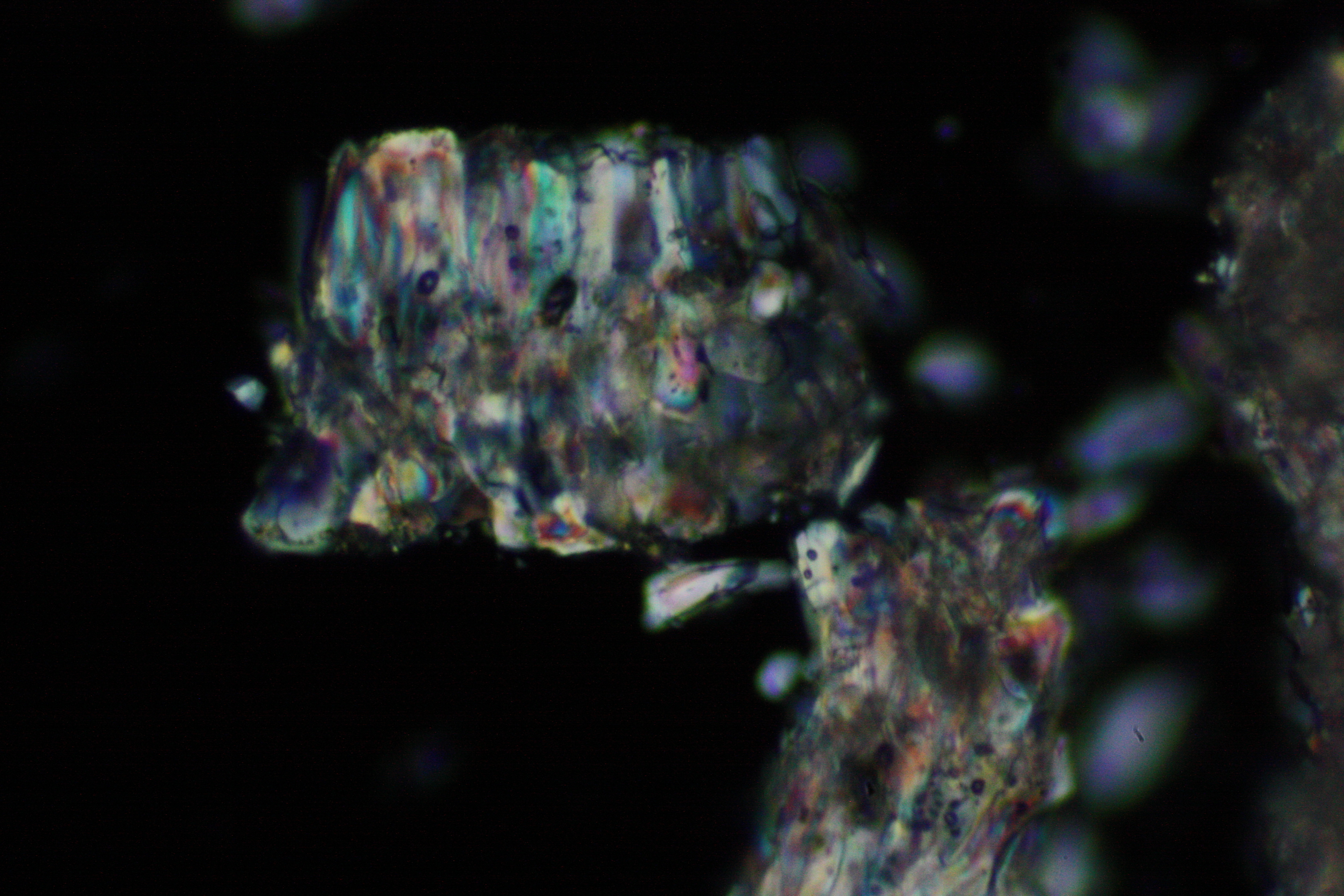
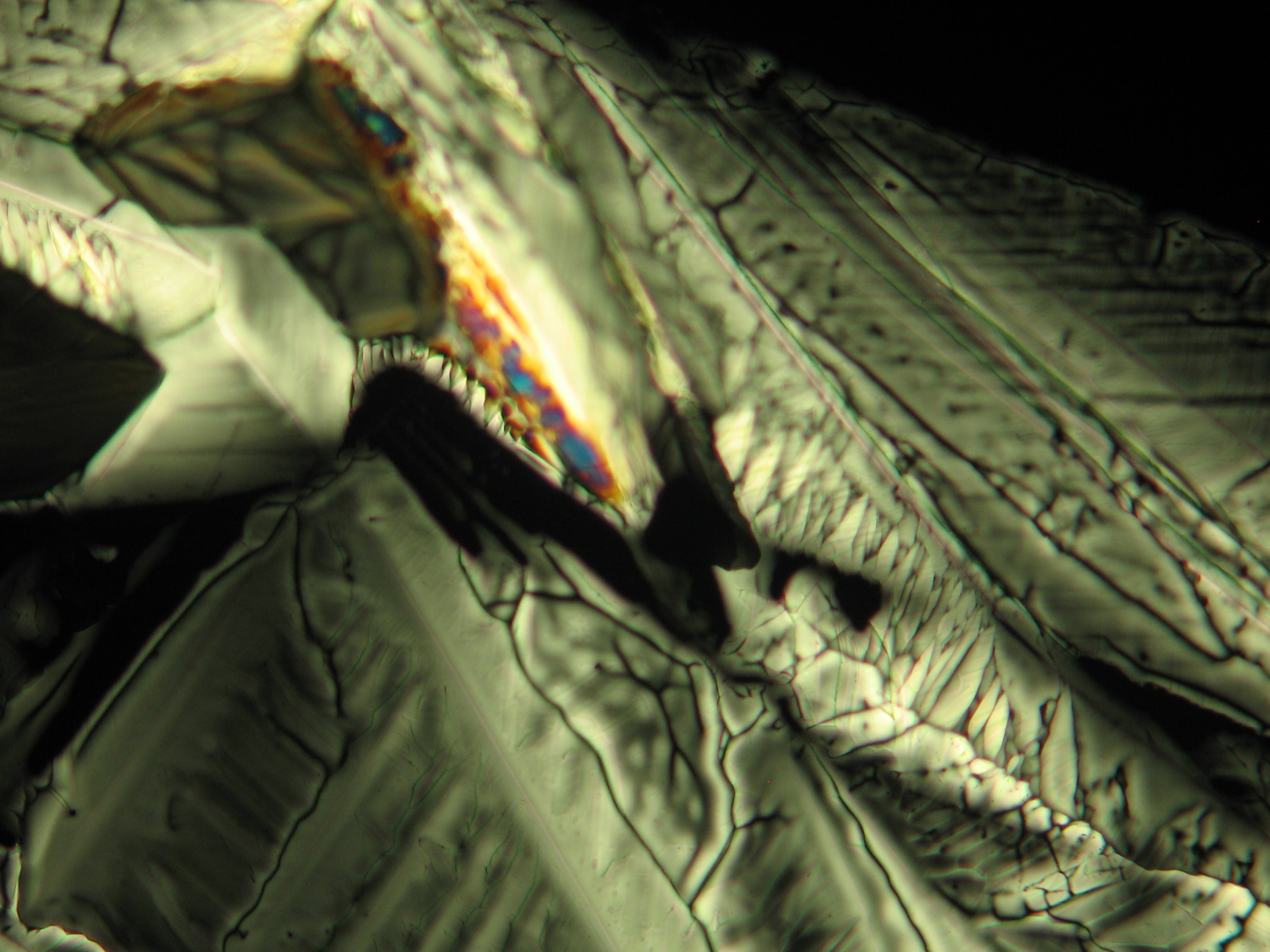
<gallery caption="Crystallized on monuments " widths="200px" heights="150px" perrow="3"> | |||
Image:0141 0010 Bb440um 0021.JPG|Sample of a salt crust of magnesium sulfate whiskers (image width 440 um) | |||
</gallery> | |||
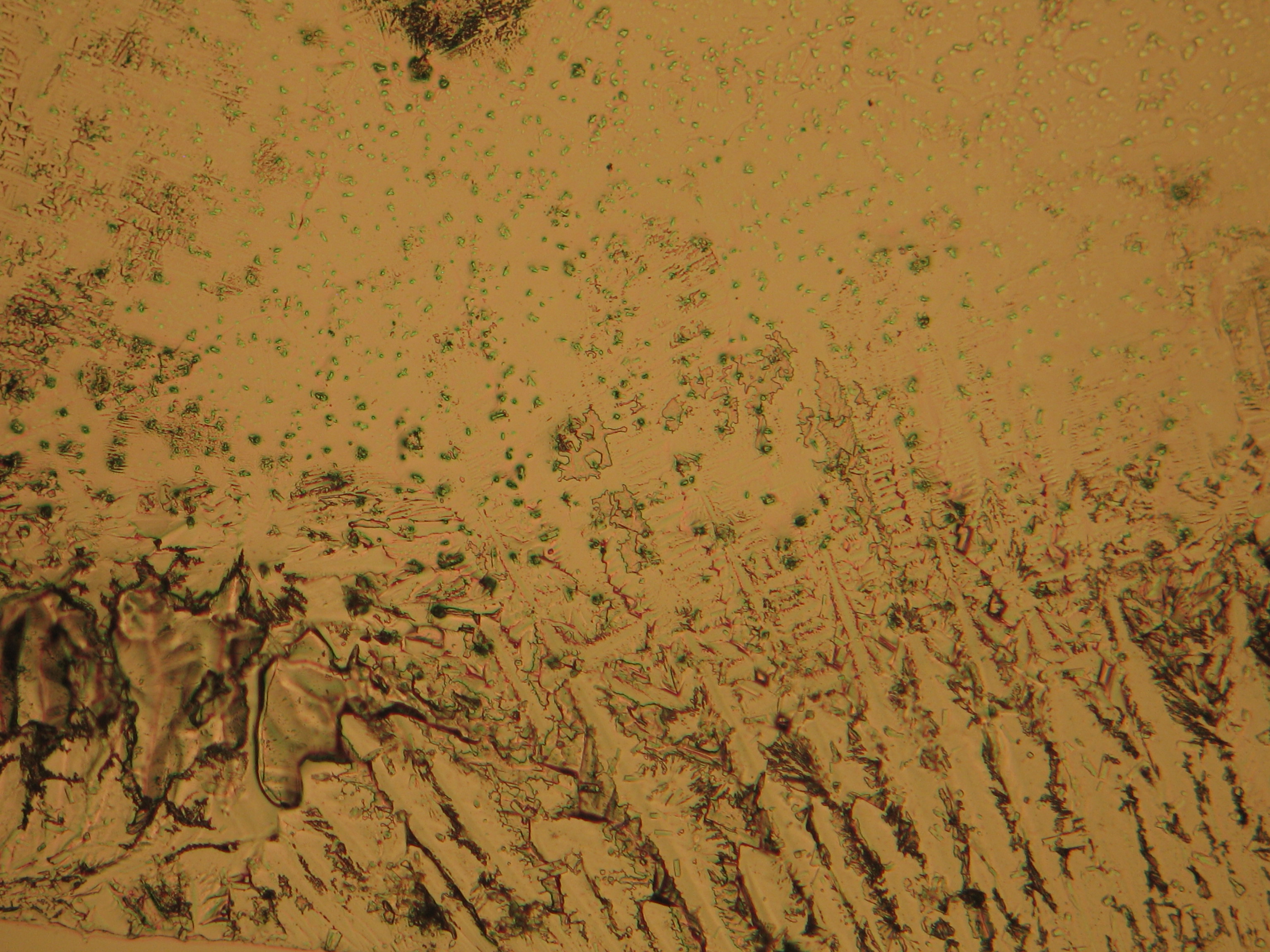
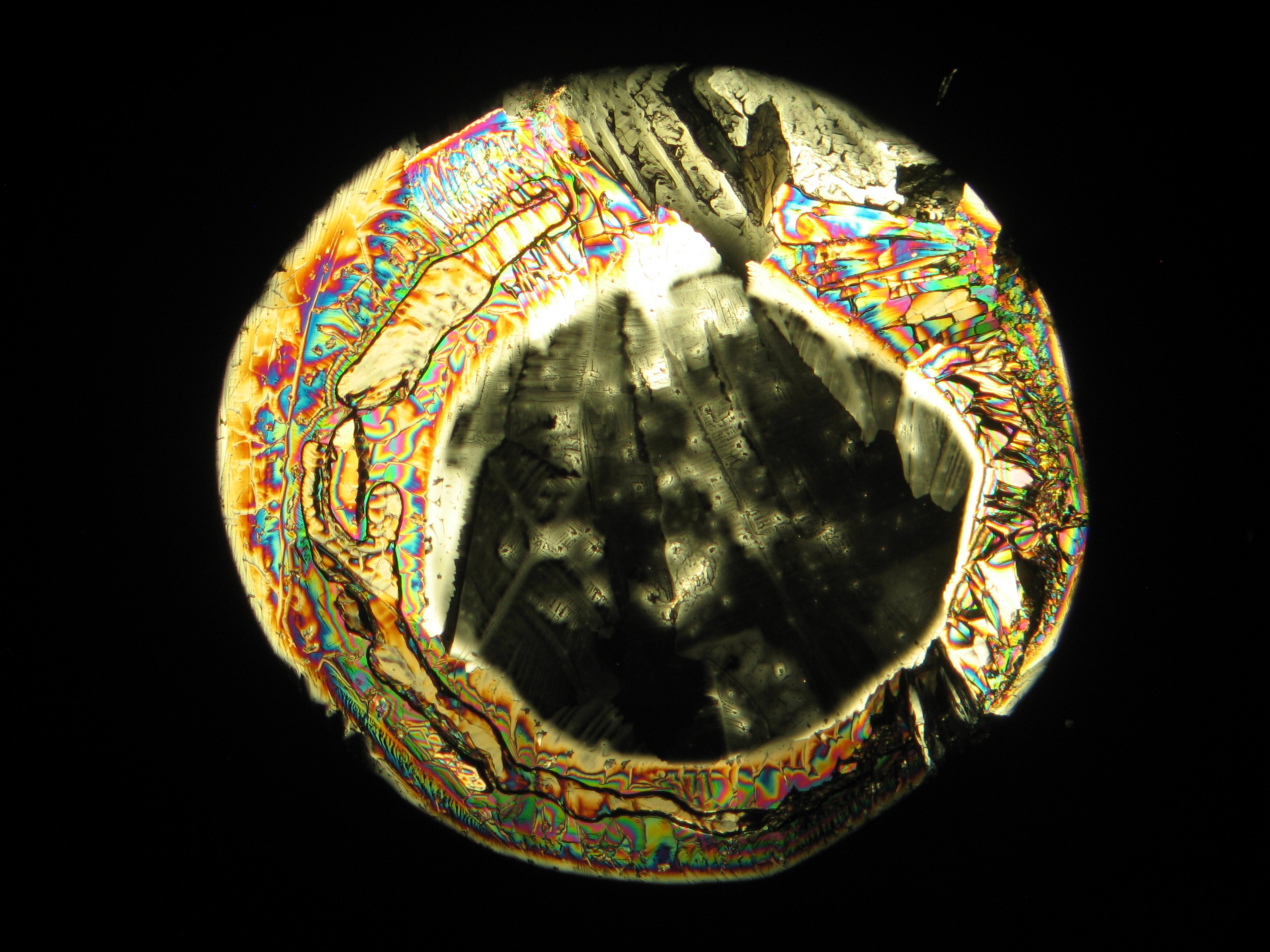
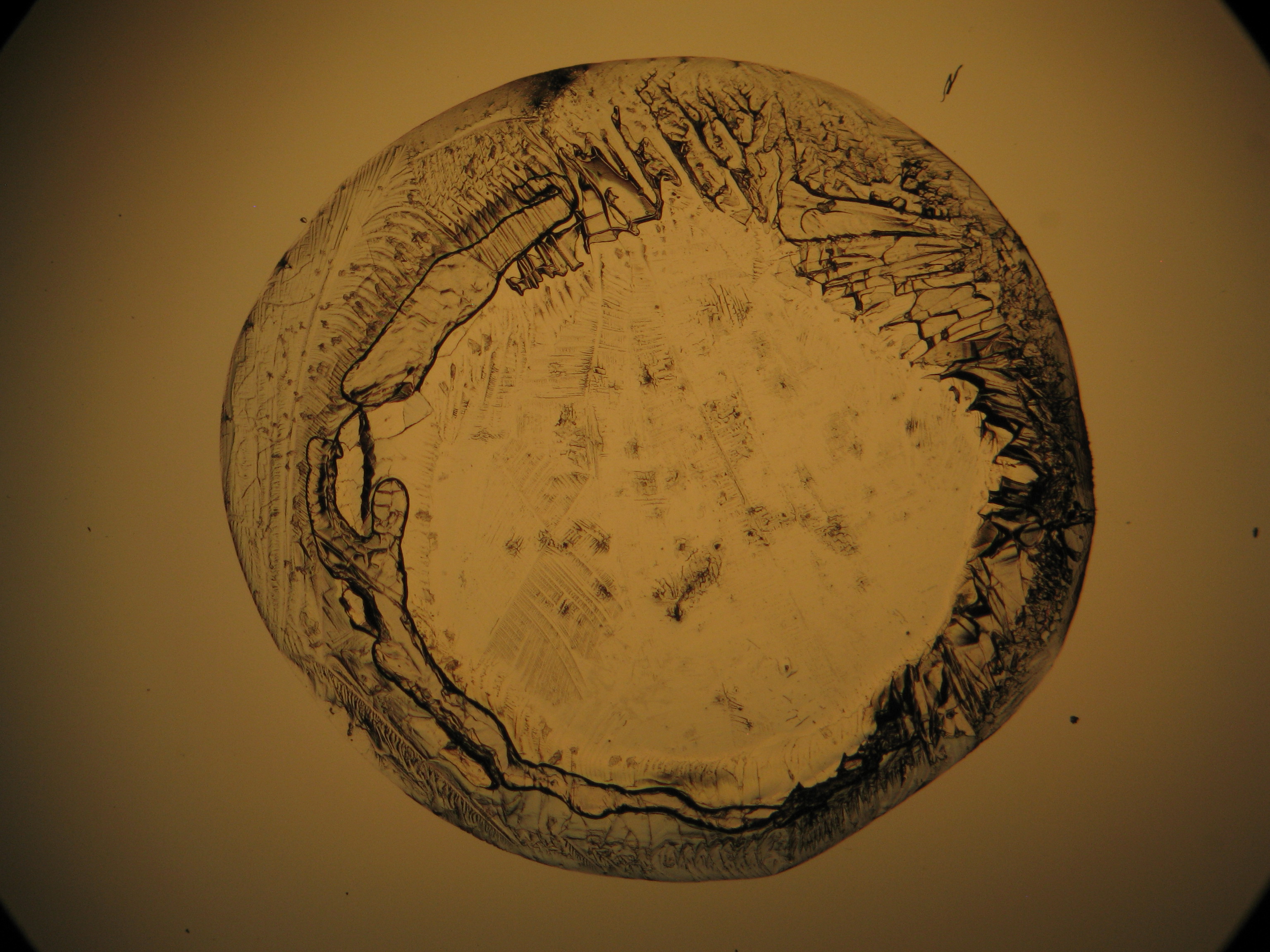
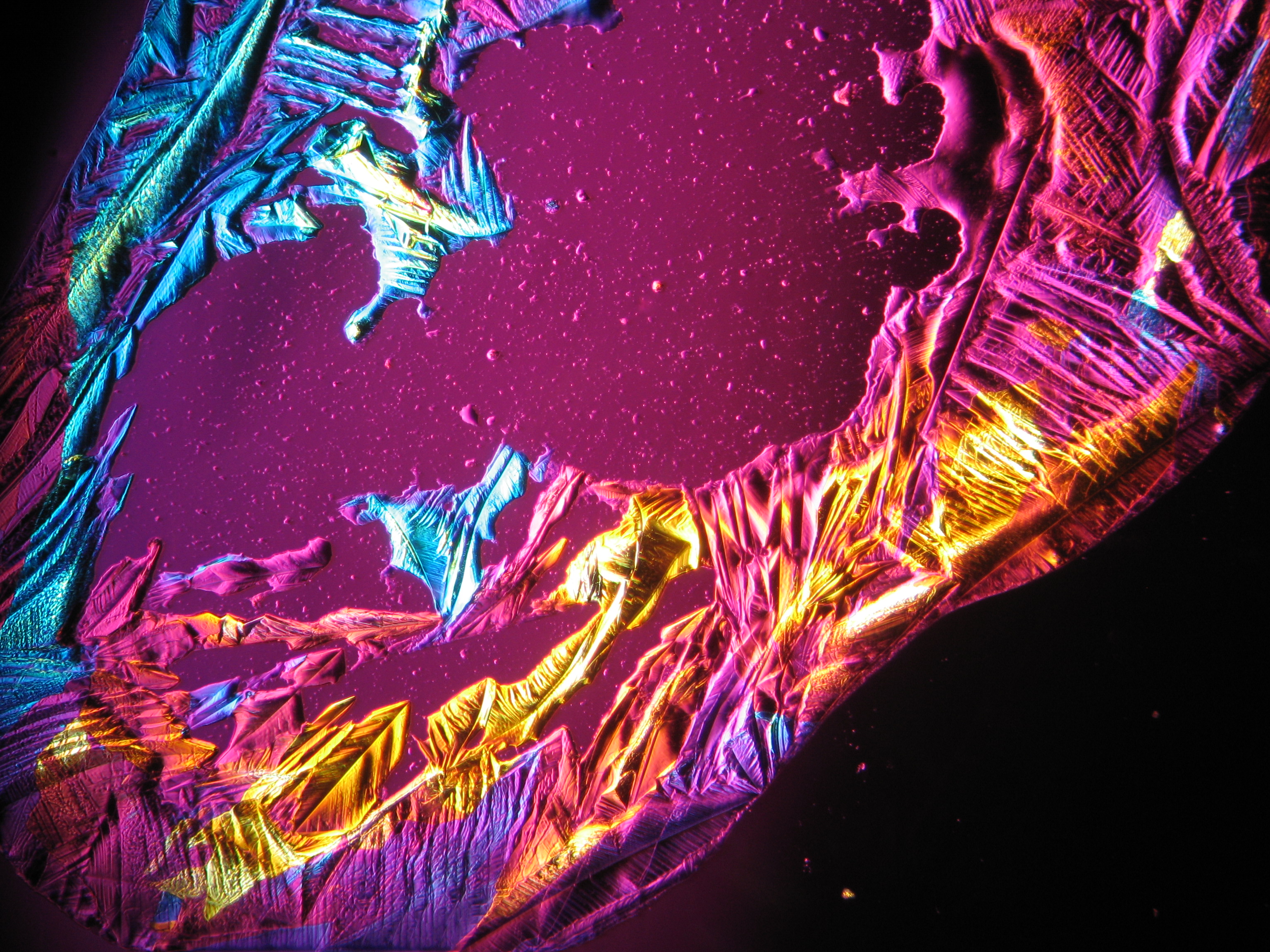
<gallery caption="Crystallized from a water solution on a glass slide " widths="200px" heights="150px" perrow="3"> | |||
Image:CeS 221209-02-10x-01.JPG|Sample of magnesium sulfate re-crystallized from water solution on a glass slide | |||
Image:CeS 221209-02-10x-02.JPG|Sample of magnesium sulfate re-crystallized from water solution on a glass slide | |||
Image:CeS 221209-04-2,5kx-01.JPG|Sample of magnesium sulfate re-crystallized from water solution on a glass slide | |||
Image:CeS 221209-04-2,5kx-02.JPG|Sample of magnesium sulfatere-crystallized from water solution on a glass slide | |||
Image:CeS 221209-04-2,5kx-03.JPG|Sample of magnesium sulfate re-crystallized from water solution on a glass slide | |||
Image:MGSO4 4.5.2006 (11).JPG|Magnesium- sulfate, crystallized from a water solution on a glass slide | |||
Image:MGSO4 4.5.2006 (12).JPG|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:MGSO4 4.5.2006 (3).JPG|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:Magnesiumsulfat pol 01.jpg|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:MgSO4 pol 400x 01.JPG|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:MgSO4 pol+analy 400x 01.JPG|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:MgSO4 pol+analy+rotII 400x 01.JPG|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
Image:MgSO4 pol+analy.jpg|Magnesium sulfate, crystallized from a water solution on a glass slide | |||
</gallery> | |||
<!-- | |||
== Unter dem Rasterelektronenmikroskop == | |||
--> | |||
==Weblinks== | |||
<references/> | |||
== Literature == | |||
<biblist/> | |||
[[Category:Epsomite]][[Category:Sulphate]][[Category:Salt]][[Category:approved]][[Category:Sulfate]][[Category:List]] | |||
Latest revision as of 10:43, 3 May 2023
Authors: Hans-Jürgen Schwarz, Tim Müller, Nils Mainusch
English Translation by Sandra Leithäuser
back to Sulfate
| Epsomite[1][2][3][4] | |

| |
| Mineralogical name | Epsomite |
| Chemical name | Magnesium sulfate heptahydrate |
| Trivial name | Bitter Salts, reichardtite, seelandite |
| Chemical formula | MgSO4•7H2O |
| Other forms | Kieserite (MgSO4•H2O) Sanderite (MgSO4•2H2O) Starkeyite (MgSO4•4H2O) Pentahydrite(MgSO4•5H2O) Hexahydrite (MgSO4•6H2O) Meridianiite (MgSO4•11H2O) Magnesium 12-Hydrate |
| Crystal system | orthorhombic |
| Crystal structure | |
| Deliquescence humidity 20°C | 91.3 % |
| Solubility (g/l) at 20°C | 2.893 mol/kg |
| Density (g/cm³) | 1.68 g/cm3 |
| Molar volume | 146.8 cm3/mol |
| Molar weight | 246.48 g/mol |
| Transparency | transparent to translucent |
| Cleavage | clear to perfect |
| Crystal habit | small, acicular, fibre like crystals, granular agregates, crusts |
| Twinning | rare |
| Phase transition | |
| Chemical behavior | |
| Comments | can be produced from an aqueous solution under 50°C |
| Crystal Optics | |
| Refractive Indices | nx = 1.432 ny = 1.453 nz = 1.4609 |
| Birefringence | Δ = 0.0284 |
| Optical Orientation | biaxial negative |
| Pleochroism | none |
| Dispersion | 51.1° |
| Used Literature | |
| [Steiger.etal:2011]Title: Weathering and Deterioration Author: Steiger, M.; Charola, A. E.; Sterflinger, K.  [Lide:1995]Title: CRC Handbook of Chemistry and Physics [Lide:1995]Title: CRC Handbook of Chemistry and PhysicsAuthor: Lide D.R.  [Dana:1951]Title: Dana's System of Mineralogy [Dana:1951]Title: Dana's System of MineralogyAuthor: Dana J.D. 
| |
Introduction[edit]
Occurrence of epsomite[edit]
The natural mineral epsomite was first extracted in 1695, from the mineral waters near Epsom (London), where the mineral name was derived from. Just as kieserite (magnesium sulfate mono hydrate), single crystals rarely occur in nature. Both magnesium salts, as well as their hydrate forms are extracted from alpine deposits and occur together with sylvite and rock salt in north Germany, and in the top layer of salt deposits by Staßfurt (near Magdeburg).
Information on the origin and formation of epsomite on monuments[edit]
The formation of magnesium sulfate on monuments requires the presence of soluble magnesium compounds, and magnesium ions. Different building materials on historic structures can contain magnesium compounds. Some examples are listed below:
- Utilization of lime with dolomite components: Dolomite (CaMg(CO3)2 ) is a double salt. If lime stone containing dolomite components is heated, slaked and used as mortar, CaCO3 and MgCO3·xH2O form through this process (e.g. Nesquehonite MgCO3·3H2O including the molecules from the water of crystallization). The water solubility of MgCO3·3H2O at 1,76 g/l, is significantly above the solubility of calcite (0,014 g/l) and dolomite (0,078 g/l). If magnesium ions have been dissolved by moisture, they can form several different magnesium salts in the presents of the corresponding anions. The formation of magnesium sulfate can occur if, in connection with dolomite containing plasters, gypsum was used for stucco or plaster, readily supplying sulfates.
- Utilization of magnesia containing binder: Magnesia binder consists essentially of MgO and MgCl2 or magnesium sulfate. The fully cured and solidified binder can contain highly hygroscopic magnesium salts, which can be converted to magnesium sulfate.
- Utilization of cement: According to the German standard, DIN 1164 [DIN1164:1994]Title: DIN 1164-1: Zement Teil 1, Zusammensetzung und Anforderungen
Author: DIN a maximum content MgO of 5 M.% is tolerated in cements. Apart from the possibility of causing damage by delayed quenching, due to the expansion of the magnesia, magnesium ions can theoretically be released and cause the formation of magnesium sulfate.
a maximum content MgO of 5 M.% is tolerated in cements. Apart from the possibility of causing damage by delayed quenching, due to the expansion of the magnesia, magnesium ions can theoretically be released and cause the formation of magnesium sulfate.
Deicing salt can be another source of magnesium sulfate, it often contains a small proportion of readily soluble MgCl2. Furthermore, the magnesium supply can occur through constant soil leaching in connection with rising damp.
Solubility properties[edit]
Author: Steiger, Michael; Linnow, Kirsten; Ehrhardt, Dorothee; Rohde, Mandy
 .
.
The solubility of the above magnesium sulfate forms is significantly above 100 g/l (20°C), and they are part of the group of easily soluble salts. In principle, this leads to a great mobility of the salt and frequent shifting of the accumulation zones inside the material fabric. Due to the influence of temperature on solubility, there is a risk of precipitation of the dissolved salts in combination with rapid temperature drops.
Hygroscopicity[edit]
The low hygroscopicity of the pure salt epsomite which becomes evident in the high value of the equilibrium moisture content in the range of 88-90% RH, cannot be considered in isolation. In mixed systems, i.e. under the influence of foreign ions the sorption point is lower (see table). The possibility of hygroscopic moisture uptake with the connected difficulties is still possible despite the high Deliquescence point.
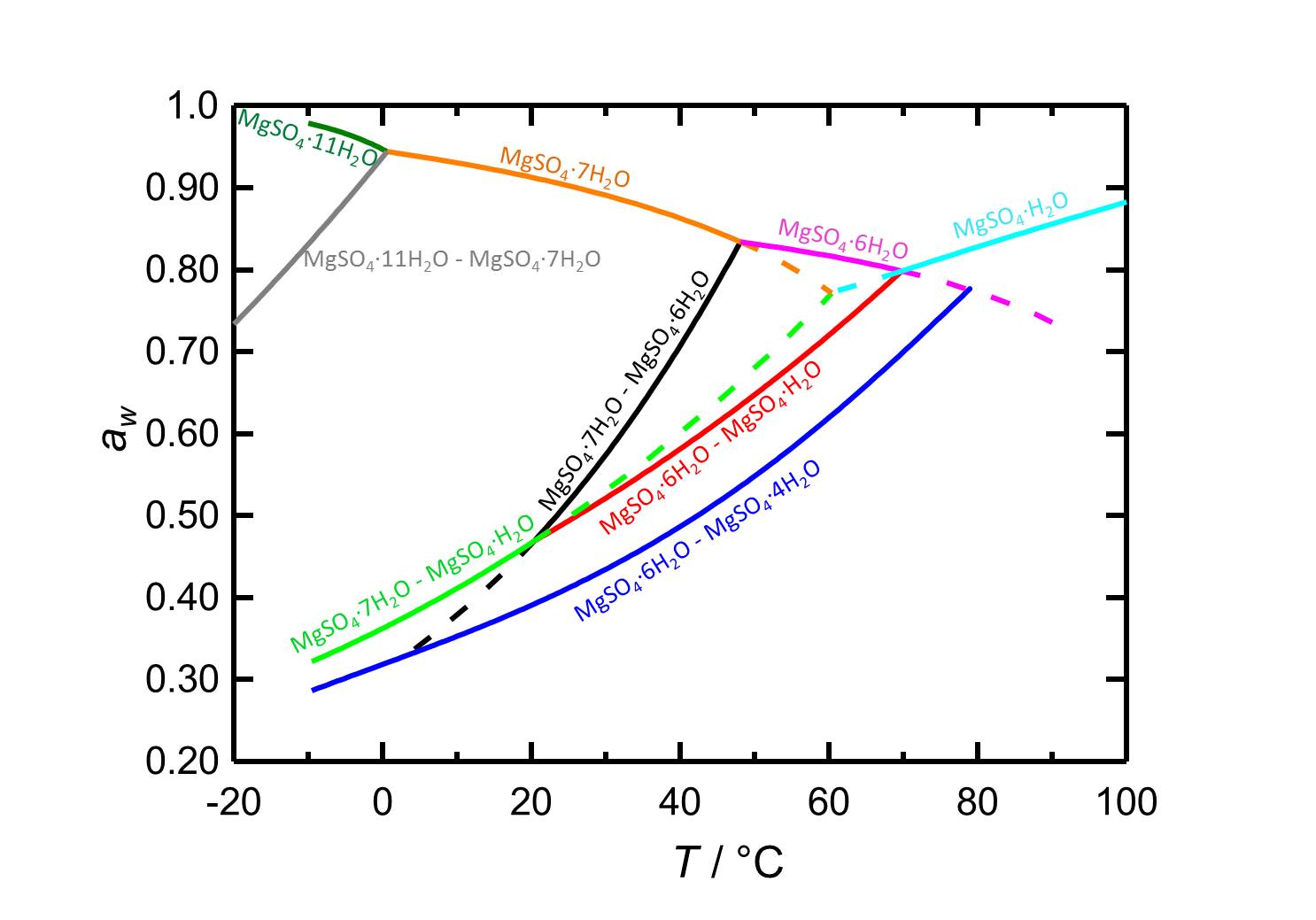
Author: Steiger, Michael; Linnow, Kirsten; Ehrhardt, Dorothee; Rohde, Mandy
 .
.
| 10°C | 20°C | 25°C | 30°C |
| 93.7% r.h. | 91.3% r.h. | 90.3% r.h. | 89.1% r.h. |
Moisture Sorption[edit]
| Moisture sorption at | 87%r.F. | 81%r.F. | 70%r.F. | 61%r.F. | 50%r.F. |
| MgSO4 | 76 | 75 | 70 | 71 | 27 |
| MgSO4 + NaCl (1:1 molar mixture) |
240 | 146 | 75 | 50 | 20 |
Crystallization pressure[edit]
Due to the high solubility of the salt, dissolving and recrystallization processes take place under the appropriate humidity conditions. In theory the occurring crystallization pressure can be calculated and ranges between 10,5-12,5 N/mm2 for epsomite and between 11,8- 14,1 N/mm2 for hexahydrite. Compared to other damaging salts these values are thus to be found in the lower third of a calculated scale of values, alltogether ranging from 7,2 bis 65,4 N/mm2 [according to [Winkler:1975]Title: Stone: Properties, Durability in Man´s Environment
Author: Winkler, Erhard M. ].
].
Hydration behavior[edit]
The system MgSO4 – H2O: the listed hydrate stages of magnesium sulfates are known to be stable compounds. With the exception of magnesium sulfate- 12- hydrate, all the above stages of the chemically combined water of magnesium sulfate on monuments are visible, but essentially only epsomite, hexahydrite, pentahydrite and kieserite occur.
At room temperature and an RH between 50%-90%, epsomite is the most stable hydrate stage. If the RH drops significantly lower than 50% at room temperature, the chemically combined water is released and lower hydrate stages are formed. Hexahydrite (MgSO4 × 6H2O) as pure salt is only stable at a temperature range of approx. 48°C to 67,5 °C. Pentahydrite has been thought to be metastable or unstable when exposed to the air, nevertheless the existence of these two salt phases has been detected by X-ray diffraction on building structures. The expulsion of the hydrate water can occur up to the formation of kieserite at elevated temperatures.
Hydration pressure[edit]
Possible variations in magnesium sulfate´s content of chemically combined water on constructions are a fact, and it is likely, that changes in the hydrate stages pentahydrite, hexahydrite and epsomite (subject to climatic fluctuations) are taking place in situ. The addition of a water molecule into the crystal lattice of hexahydrite, resulting in the transformation to epsomite, is associated with an increase in volume of around 10%. At temperatures from 0-20°C and 70% RH the resulting hydration pressure can be specified at values between 6,8 –9,7 N/mm2. The conversion from kieserite to hexahydrite causes an increase in volume of approx. 140% [according to [Stark.etal:1996]Title: Bauschädliche Salze
Author: Stark, Jochen; Stürmer, Sylvia ].
].
Conversion reaction[edit]
The following case is a good example for the potential damage connected with the formation of epsomite: if magnesium carbonate (as magnesite) is present in the material structure of a monument, epsomite can form through the influence of sulfuric acid. According to [Stark.etal:1996]Title: Bauschädliche Salze
Author: Stark, Jochen; Stürmer, Sylvia this conversion is linked to an increase in volume of over 400%.
this conversion is linked to an increase in volume of over 400%.
Analytical identification[edit]
On objects, crystallized magnesium sulfate can appear in differing morphologies, but specific forms appear more frequently. One examined object displayed a loose crust of an opaque, gray to yellow substance in situ, which was a mixture of epsomite and hexahydrite (part of the thesis by Mainusch [Mainusch:2001]Title: Erstellung einer Materialsammlung zur qualitativen Bestimmung bauschädlicher Salze für Fachleute der Restaurierung
Author: Mainusch, Nils ). At the monastery church St. Johann at Müstair epsomite appeared as “granular crusts”. Magnesium sulfate efflorescence in the shape of salt whiskers was detected at St. Georgs church in Steiermark (Styria).
). At the monastery church St. Johann at Müstair epsomite appeared as “granular crusts”. Magnesium sulfate efflorescence in the shape of salt whiskers was detected at St. Georgs church in Steiermark (Styria).
Microscopy[edit]
Laboratory examination:
Breathing onto a (predominantly) magnesium sulfate efflorescence, causes no observable macroscopic changes. However, characteristics are: the good solubility in water, a pH of 7, the formation of a ring-shaped slightly raised seam (visible by the naked eye), or a transparent layer that remains in place after the evaporation of the aqueous solvent.
In laboratory tests on objects the results should be double checked with the microscope. Observations of the raw sample material and the recrystallized salt, show that magnesium sulfate crystals appear in unspecific forms in sample material.
Experiments to produce well-shaped, single magnesium sulfate crystals, by recrystallization in aqueous solution have not been very successful, because of a strong tendency for intergrowth. Usually the previously mentioned, distinct, ring-shaped seam of intergrown crystals forms, when the magnesium sulfate containing solute is evaporated. Epsomite shows a low solubility in anhydrous ethanol and glycerin under the microscope.
For further examinations using polarized light microscopy, a considerable difficulty is the creation of good preparations, due to the tendency of magnesium sulfate to form only few equidimensional or elongated single crystals. The low solubility in ethanol, nevertheless allows to isolate single particles by repeatedly adding recrystallized material. Both on the base material as well as the recrystallized product, examination using polarized light microscopy should be carried out.
Refractive indices: nX = 1,433, ny = 1,455, nz
Birefringence: Δ = max. 0,028
Crystal class: orthorhombic
Polarized light microscopic examination:
The allocation of epsomite to the refractive indices is carried out according to the immersion method (successively by means of embedding media nD=1,518; nD=1,47; nD=1,46). When using an immersion medium with a refractive index of nD=1,45 on many single particles a slight but distinct variation in relief is visible at rotation. Because epsomite belongs to the class of orthorhombic crystals, it never appears in an oblique but always in a parallel symmetrical extinction. Beside epsomite, hexahydrite often forms, which is monoclinic and has nearly identical refractive indices. A distinction between these two hydrate stages of magnesium sulfate is only possible through the allocation of the crystal class. Due to the low path difference epsomite crystals usually only show low inference colors in the range of the first order.
Possibility for mistakes:
Epsomite/hexahydrite are to be allocated, once the examination criteria below have been clarified:
- good water solubility
- characteristic appearance at recrystallization
- low birefringence
Salts and deterioration pattern[edit]
On objects[edit]
Under the polarising microscope[edit]
- Crystallized on monuments
- Crystallized from a water solution on a glass slide
Weblinks[edit]
- ↑ http://webmineral.com/data/Epsomite.shtml seen on 29.072010
- ↑ http://www.mindat.org/min-1393.html seen on 29.072010
- ↑ <bib id="Steiger.etal:2011">
- ↑ <bib id="Lide:1995">
Literature[edit]
| [DIN1164:1994] | DIN (1994): DIN 1164-1: Zement Teil 1, Zusammensetzung und Anforderungen. |  |
| [Dana:1951] | Dana E.S. (eds.) Dana J.D. (1951): Dana's System of Mineralogy, 7, Wiley & Sons |  |
| [Lide:1995] | Lide D.R. (eds.) Lide D.R. (1995): CRC Handbook of Chemistry and Physics, CRC Press |  |
| [Mainusch:2001] | Mainusch, Nils (2001): Erstellung einer Materialsammlung zur qualitativen Bestimmung bauschädlicher Salze für Fachleute der Restaurierung, Diplomarbeit, HAWK Hochschule für angewandte Wissenschaft und Kunst Hildesheim/Holzminden/Göttingen, file:Diplomarbeit Nils Mainusch.pdf |   |
| [Stark.etal:1996] | Stark, Jochen; Stürmer, Sylvia (1996): Bauschädliche Salze, Bauhaus-Univ. Weimar |  |
| [Steiger.etal:2011] | Steiger, M.; Charola, A. E.; Sterflinger, K. (2011): Weathering and Deterioration. In: Siegesmund, Siegfried; Snethlage, Rolf (eds.): Stone in Architecture - Properties, Durability, Springer, 227-316. |  |
| [Steiger.etal:2011a] | Steiger, Michael; Linnow, Kirsten; Ehrhardt, Dorothee; Rohde, Mandy (2011): Decomposition reactions of magnesium sulfate hydrates and phase equilibria in the MgSO4-H2O and Na+-Mg2+-Cl--SO42--H2O systems with implications for Mars. In: Geochimica et Cosmochimica Act, 75 (12), 3600-3626, 10.1016/j.gca.2011.03.038, |  |
| [Vogt.etal:1993] | Vogt, R.; Goretzki, Lothar (1993): Der Einfluss hygroskopischer Salze auf die Gleichgewichtsfeuchte und Trocknung anorganischer Baustoffe, unveröffentlichter Bericht. |  |
| [Winkler:1975] | Winkler, Erhard M. (1975): Stone: Properties, Durability in Man´s Environment, Springer Verlag, Wien |  |